It is a radioactive material created by bombarding stable phosphorus with neutrons in a controlled environment. β- (1.71 MeV) particle in 32S with a half-life of 14.263 days is emitted as a byproduct of its disintegration. When it comes to phosphorus isotopes, phosphorus 32 (P-32) is the one whose nucleus has 15 protons and 17 neutrons.
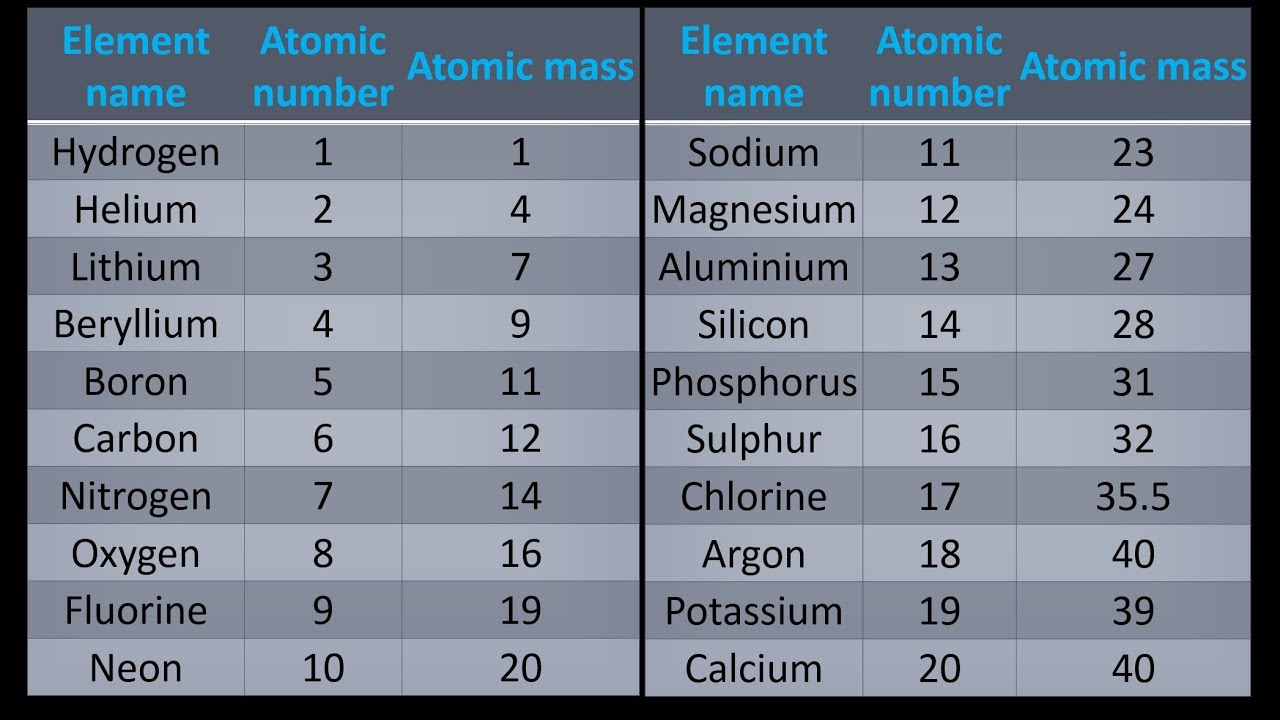
All other isotopes have a half-life of fewer than 2.5 minutes, and the vast majority of them have a half-life of less than one second, as well. Phosphorus 33 (P-33) and phosphorus 32 (P-32) are the radioisotopes of phosphorus with the longest half-lives (14.263 days). Phosphorus 33 (P-33) has a half-life of 25.34 days, while phosphorus 32 (P-32) has a half-life of 25.34 days. Phosphorus is a monoisotopic element since there is only one stable isotope, P-31. With a mass number spanning from 24 to 46, phosphorus (P) has 23 known isotopes, each with a different mass number. The least stable is 25P, which has a half-life of fewer than 30 nanoseconds and is the least stable. All others have half-lives of less than 2.5 minutes, with the majority of them being less than a second. The radioactive isotopes with the longest half-lives are 33P, which has a half-life of 25.34 days, and 32P, which has a half-life of 14.268 days. Isotopes of phosphorusĭespite the fact that phosphorus (15P) has 23 stable isotopes ranging from 25P to 47P, only 31P is stable, and as a result, phosphorus is classified as a monoisotopic element. It is the weighted average of the atomic masses of the element’s isotopes on a scale where the carbon-12 atom has an exact mass of 12 units that is used to calculate its relative atomic mass. When it comes to elements with only one naturally occurring isotope, such as beryllium or fluorine, the atomic mass is equal to the atomic weight of the element. In this case, it is a unit-less figure that represents the relationship between atomic masses of naturally occurring isotopes of an element and the atomic mass of one-twelfth of the mass of carbon-12. The weight of an atom, in contrast to the weight of an item, has nothing to do with the force of gravitation. It has a total of twelve protons and six neutrons. The carbon-12 molecule, which is still in use as a standard today, is composed of six protons and six neutrons, with an atomic mass of twelve amu. The electrons in an atom have such a small mass when compared to the protons and neutrons that their mass is virtually non-existent. The mass of each proton and neutron is approximately 1 amu (1.0073 and 1.0087, respectively). Phosphorus has an atomic mass of 30.973762 u.Ītomic mass is defined as the number of protons and neutrons in an atom where the mass of each proton and neutron is about 1 amu. Phosphorus is most commonly found in the form of phosphate in minerals. This element has a concentration of around one gramme per kilogramme of the Earth’s crust (compare copper at about 0.06 grams). However, due to the extreme reactivity of phosphorus, it is never found as a free element on the surface of the Earth. Elemental phosphorus comes in two major forms: white phosphorus and red phosphorus. Phosphorus is a chemical element with the symbol P and the atomic number 15.
